Search Results
258 results found with an empty search
- Bayer-Sponsored Educational Sessions at Imaging in Japan 2026
We are pleased to announce two special industry-supported educational sessions sponsored by our Gold Sponsor Bayer during Imaging in Japan 2026, hosted at the beautiful Hotel Chinzanso Tokyo. These sessions will provide valuable opportunities for learning, discussion, and professional engagement with leaders in radiology. Bayer Lunch & Learn Date & Time: Monday, June 1, 2026 · 12:00 PM – 1:00 PM Location: ZUIKO Room, Hotel Chinzanso Tokyo Moderator: Neil Rofsky This Bayer-sponsored Lunch & Learn session will be open to all registered conference attendees and will feature an educational presentation and discussion led by distinguished faculty. Attendees will have the opportunity to learn about emerging developments in imaging and participate in an interactive dialogue with experts in the field. BAYER AG Bayer Focus Group Discussion Date & Time: Wednesday, June 3, 2026 · 12:00 PM – 1:00 PM Location: TSUZUMI Room, Hotel Chinzanso Tokyo Moderator: Neil Rofsky In addition to the Lunch & Learn, Bayer will host an exclusive focus group discussion bringing together approximately 12 invited radiologists for a moderated roundtable conversation. This small-group format is designed to encourage open dialogue and exchange of professional insights among key clinicians. These sponsored sessions reflect the important collaboration between industry innovators and the global radiology community, helping advance education, dialogue, and innovation in medical imaging. We thank Bayer for its support of Imaging in Japan 2026 and look forward to welcoming participants to these engaging events in Tokyo.
- Bilateral Facet Dislocation at C7-T1
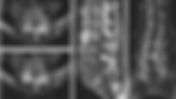
A 70-year-old female fell off her bed while changing a light bulb and complains of left shoulder pain. What is the diagnosis? • Xray of the Week Figure 1. Non-contrast CT of the cervical spine. What is the diagnosis? Figure 2. Non-contrast CT of the cervical spine (sagittal reconstructions): Bilateral facet dislocation at C7-T1 with anterior subluxation of C7 on T1. The inferior facets of C7 are locked anterior to the superior facets of T1 (yellow and green arrows), with anterior vertebral body translation present. There are associated fractures of C7 and T1 (red arrows). Figure 2. Non-contrast axial CT of the cervical spine: Bilateral facet dislocation at C7-T1 with naked facet sign (green arrow) and reverse hamburger bun sign (red arrow). Discussion The cervicothoracic junction (C7–T1) represents a biomechanical transition between the mobile cervical spine and the rigid thoracic spine, making it particularly vulnerable to traumatic instability. Bilateral facet dislocation at this level is a highly unstable injury most commonly associated with high-energy trauma but may also occur after low-energy falls in elderly patients due to osteopenia and degenerative stiffness. [1] The injury involves both facets "jumping" anteriorly, disrupting ligamentous stability and risking cord compression.[2] Delayed diagnosis can occur due to poor visualization of C7-T1 on plain films, emphasizing the need for CT in suspected cases.[3] As seen in this case, "teardrop" fragment on C7 or T1 represents a highly unstable flexion-compression or flexion-distraction injury; it is not a minor avulsion and signifies severe ligamentous damage.[5] Imaging Findings CT is the imaging modality of choice for evaluating suspected cervicothoracic junction trauma, as plain radiographs frequently fail to visualize C7–T1 due to shoulder overlap. Multiplanar CT reformations are essential for assessing alignment, facet integrity, and associated fractures. [1] Key CT findings include: High-grade anterior translation of C7 relative to T1 Bilateral jumped or locked facets with loss of normal facet articulation Widening of the interspinous distance indicating posterior ligamentous complex disruption Small anterior vertebral body fracture fragments at C7 and T1 consistent with flexion teardrop fractures Marked narrowing of the spinal canal at the C7–T1 level Axial CT images demonstrate bilateral reverse hamburger bun and naked facet signs, which are pathognomonic for facet dislocation. [2,3] Differential Diagnosis Traumatic bilateral facet dislocation must be distinguished from degenerative spondylolisthesis, which is common in geriatric patients but lacks joint capsule disruption and typically involves less than 4 mm of translation. Unilateral facet dislocation involves a rotational component and manifests as a "bow-tie" sign on lateral radiographs with less than 25% anterior translation. Isolated flexion teardrop fractures involve significant three-column injury but lack the mechanical "locking" of the facet joints seen in dislocations. Traumatic dislocation is favored when there is facet joint disruption, significant translation, and associated ligamentous injury. [1] Management and Prognosis Bilateral facet dislocation at C7–T1 represents a surgical emergency due to profound instability and high risk of neurologic deterioration. Initial management includes cervical immobilization and urgent neurosurgical consultation. Most cases require operative fixation, often via posterior or combined anterior-posterior approaches. Prognosis depends largely on neurologic status at presentation. [1] Key Learning Points C7–T1 injuries are frequently missed on plain radiographs Low-energy falls can cause severe cervical instability in elderly patients Reverse hamburger bun and naked facet signs are key CT indicators Anterior teardrop fractures signify severe ligamentous injury Early recognition and surgical management are critical Correlate with MRI for soft tissue/cord assessment. References Raniga SB, Menon V, Al Muzahmi KS, Butt S. MDCT of acute subaxial cervical spine trauma: a mechanism-based approach. Insights Imaging. 2014;5(3):321–338. doi: 10.1007/s13244-014-0311-y Daffner SD, Daffner RH. Computed tomography diagnosis of facet dislocations: the hamburger bun and reverse hamburger bun signs. J Emerg Med. 2002;23(4):387–394. DOI: 10.1016/s0736-4679(02)00577-2 Lingawi SS. The naked facet sign. Radiology. 2001;219(2):366–367. DOI: 10.1148/radiology.219.2.r01ma06366 Kim KS, Chen HH, Russell EJ, Rogers LF. Flexion teardrop fracture of the cervical spine: radiographic characteristics. AJR Am J Roentgenol. 1989;152(2):319–326. DOI: 10.2214/ajr.152.2.319 Kim KS, Chen HH, Russell EJ, Rogers LF. Flexion teardrop fracture of the cervical spine: radiographic characteristics. AJR Am J Roentgenol . 1989;152(2):319-326. DOI: 10.2214/ajr.152.2.319 Nishad Kosaraju is a second-year osteopathic medical student (OMS-II) at the Edward Via College of Osteopathic Medicine–Carolinas Campus ( VCOM-Carolinas ). He has a strong interest in diagnostic imaging and interventional radiology, with particular enthusiasm for case-based learning and understanding how imaging directly informs clinical decision-making. Nishad did his undergraduate education at UNC Chapel Hill , where he studied Exercise and Sports Science. Nishad enjoys contributing educational radiology cases that highlight classic imaging findings, uncommon presentations of common conditions, and high-yield diagnostic pearls for trainees. Outside of medicine, he enjoys golf, classical music, and technological advances in medicine. Kevin M. Rice, MD is the president of Global Radiology CME and is a radiologist with Cape Radiology Group . He has held several leadership positions including Board Member and Chief of Staff at Valley Presbyterian Hospital in Los Angeles, California. Dr. Rice has made several media appearances as part of his ongoing commitment to public education. Dr. Rice's passion for state-of-the-art radiology and teaching includes acting as a guest lecturer at UCLA. In 2015, Dr. Rice and Natalie Rice founded Global Radiology CME to provide innovative radiology education at exciting international destinations, with the world's foremost authorities in their field. In 2016, Dr. Rice was nominated and became a semifinalist for a "Minnie" Award for the Most Effective Radiology Educator. He was once again a semifinalist for a "Minnie" for 2021's Most Effective Radiology Educator by AuntMinnie.com . He has continued to teach by mentoring medical students interested in radiology . Everyone he has mentored has been accepted into top programs across the country, including Harvard, UC San Diego, Northwestern, Vanderbilt, and Thomas Jefferson. Follow Dr. Rice on Twitter @KevinRiceMD All posts by Kevin M. Rice, MD
- Methamphetamine Associated Cardiomyopathy
34 year old male with chest pain and shortness of breath • Xray of the Week Figure 1. 34 year old male with chest pain and shortness of breath. Figure 2. A and B: Axial and coronal CT with cardiomegaly due to markedly dilated heart. Right pleural effusion (blue arrow). Low EF with contrast only in RA (red arrows) and RV (orange arrow). Reflux of contrast into the IVC (green arrow) and hepatic veins (white arrow) indicates tricuspid valve regurgitation. C: Echocardiogram apical 4 chamber view. Mitral regurgitation as evidenced by a regurgitant jet extending into the RA (yellow arrow). Discussion: Methamphetamine (MA) and related compounds are the most widely abused drugs in the world after cannabis. It is a psychostimulant that causes an increase in the synapse of monoamine neurotransmitters, including dopamine, norepinephrine, and serotonin [1]. Methamphetamines can be smoked, snorted, injected, or ingested orally. Methamphetamine is more potent, and its effects last longer than cocaine. Methamphetamine associated cardiomyopathy (MACM) is more common in younger age groups compared to patients with cardiomyopathy attributable to other causes. The development of MACM has been shown to be dose-dependent and amplified by repetitive use, binge pattern use, and concomitant use of other substances. Autopsy studies have shown MACM to be associated with extensive myocardial fibrosis, cellular vacuolization, and myocyte destruction [2, 3]. Cardiac complications of MA use include chest pain, hypertension, arrhythmia, aortic dissection, coronary vasospasm, cardiomyopathy, sudden cardiac death, and pulmonary arterial hypertension [2]. Figure 3. Axial (A) and coronal (B) contrast-enhanced CT images in the same patient demonstrate a dilated right ventricle containing a non-enhancing filling defect, consistent with a right ventricular thrombus. (red arrows). Imaging Findings Echocardiography typically shows severe multi-chamber dilatation, reduced ejection fraction (EF), mitral regurgitation (MR), tricuspid regurgitation (TR), and pericardial effusion [6-9]. Patients with MACM are also prone to developing intracardiac thrombi, with up to 33% for LV thrombus and 3.3% for RV thrombus [4, 5] (Fig. 3), with RV thrombi posing a significant risk for pulmonary embolism. Thrombus formation is driven by the triad of severe systolic stasis, drug-induced endocardial injury, and a prothrombotic state. [5]. In general, patients with MACM have significantly larger LA, LV, and RV size, lower LVEF, and a higher rate of mitral regurgitation (MR) compared to other causes of dilated cardiomyopathy [6]. Contrast-enhanced CT typically demonstrates severe biventricular dilatation and associated findings of pulmonary edema like pleural effusions or septal thickening. Contrast-enhanced CT may show a hallmark sign of severe tricuspid regurgitation: the reflux of intravenous contrast into the inferior vena cava and hepatic veins. Additionally, CT can help exclude other stimulant-related complications like aortic dissection or coronary artery calcification. Cardiac Magnetic Resonance (CMR) remains the gold standard for tissue characterization, identifying linear mid-wall septal fibrosis, but its use is often limited in the acute phase. On CMR, the presence of late gadolinium enhancement (LGE) is a key marker of irreversible myocardial damage and predicts poor functional recovery. In this case, there is severe tricuspid regurgitation (TR) with reflux of contrast into the inferior vena cava (IVC) and hepatic veins; pleural effusion is indicative of heart failure (Figs. 2 A, B). There is also MR visualized on the echocardiogram with a large regurgitant jet extending into the LA during systole (Fig. 2C). Differential Diagnosis MACM must be distinguished from idiopathic dilated cardiomyopathy, which typically occurs in older demographics and lacks the same potential for reversibility. Alcoholic cardiomyopathy presents similarly, but right heart involvement and intracardiac thrombus are often more pronounced in methamphetamine users. Viral myocarditis should also be considered, though it frequently presents with sub-epicardial LGE rather than the mid-wall or sub-endocardial patterns seen in MACM. Management and Prognosis The mainstay of management is complete drug abstinence, which can lead to dramatic structural and functional recovery. Treatment of MACM is aimed at the specific pathology such as anticoagulation for intracardiac thrombus and diuresis/venodilators for volume overload. Prognosis is largely dictated by the extent of myocardial fibrosis present at the time of diagnosis. Key Learning Points Suspect MACM in young patients with unexplained cardiomegaly and heart failure. CT findings include marked multi-chamber dilatation and contrast reflux into the hepatic veins. Intracardiac thrombi are highly prevalent (up to 33%) and can involve both ventricles. Cardiac MR is superior for tissue characterization and prognostication, but CT is vital in the emergency setting. Cardiac recovery is highly dependent on achieving sustained drug abstinence. References: Barr, A.M., et al., The need for speed: an update on methamphetamine addiction. J Psychiatry Neurosci, 2006. 31(5): p. 301-13. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1557685/ Reddy, P.K.V., et al., Clinical Characteristics and Management of Methamphetamine-Associated Cardiomyopathy: State-of-the-Art Review. J Am Heart Assoc, 2020. 9(11): p. e016704 DOI: 10.1161/jaha.120.016704. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7428977/ Karch, S.B., The unique histology of methamphetamine cardiomyopathy: a case report. Forensic Sci Int, 2011. 212(1-3): p. e1-4 DOI: 10.1016/j.forsciint.2011.04.028. Retrieved from https://pubmed.ncbi.nlm.nih.gov/21664081/ Janardhanan, R. and A. Kannan, Methamphetamine Cardiotoxicity: Unique Presentation with Multiple Bi-Ventricular Thrombi. The American Journal of Medicine, 2016. 129(1): p. e3-e4 DOI: 10.1016/j.amjmed.2015.08.006. Retrieved from https://www.amjmed.com/article/S0002-9343(15)00780-9/fulltext Schürer, S., et al., Clinical Characteristics, Histopathological Features, and Clinical Outcome of Methamphetamine-Associated Cardiomyopathy. JACC Heart Fail, 2017. 5(6): p. 435-445 DOI: 10.1016/j.jchf.2017.02.017. Retrieved from https://pubmed.ncbi.nlm.nih.gov/28571597/ Ito, H., et al., A comparison of echocardiographic findings in young adults with cardiomyopathy: with and without a history of methamphetamine abuse. Clin Cardiol, 2009. 32(6): p. E18-22 DOI: 10.1002/clc.20367. Retrieved from https://pubmed.ncbi.nlm.nih.gov/19330818/ Neeki, M.M., et al., Frequency of Methamphetamine Use as a Major Contributor Toward the Severity of Cardiomyopathy in Adults ≤50 Years. The American Journal of Cardiology, 2016. 118(4): p. 585-589 DOI: https://doi.org/10.1016/j.amjcard.2016.05.057. Retrieved from http://www.sciencedirect.com/science/article/pii/S0002914916309602 Wijetunga, M., et al., Crystal methamphetamine-associated cardiomyopathy: tip of the iceberg? J Toxicol Clin Toxicol, 2003. 41(7): p. 981-6 DOI: 10.1081/clt-120026521. Retrieved from https://pubmed.ncbi.nlm.nih.gov/14705845/ Yeo, K.K., et al., The association of methamphetamine use and cardiomyopathy in young patients. Am J Med, 2007. 120(2): p. 165-71 DOI: 10.1016/j.amjmed.2006.01.024. Retrieved from https://pubmed.ncbi.nlm.nih.gov/17275458/ Voskoboinik A, Ihle JF, Bloom JE, et al. Methamphetamine-associated cardiomyopathy: patterns and predictors of recovery. Intern Med J . 2016;46(6):723-727. doi:10.1111/imj.13050 https://doi.org/10.1111/imj.13050 PubMed: https://pubmed.ncbi.nlm.nih.gov/26929061/ Koo BH, et al. Recreational Drug-induced Cardiopulmonary Injury. RadioGraphics . 2025;45(10). doi:10.1148/rg.250013 https://doi.org/10.1148/rg.250013 Zuern CS, Sticherling C, Krisai P, et al. Methamphetamine-associated cardiomyopathy. Eur Heart J Cardiovasc Imaging . 2024;25(4):e147. doi:10.1093/ehjci/jead320 https://doi.org/10.1093/ehjci/jead320 PubMed: https://pubmed.ncbi.nlm.nih.gov/37983477/ Hagan IG, Burney K. Radiology of recreational drug abuse. RadioGraphics . 2007;27(4):919-940. doi:10.1148/rg.274065103 https://doi.org/10.1148/rg.274065103 PubMed: https://pubmed.ncbi.nlm.nih.gov/17620470/ Pujol-López M, Ortega-Paz L, Flores-Umanzor EJ, et al. Cardiac Magnetic Resonance as an Alternative to Endomyocardial Biopsy to Predict Recoverability of Left Ventricular Function in Methamphetamine-Associated Cardiomyopathy. JACC Heart Fail . 2017;5(11):853-854. doi:10.1016/j.jchf.2017.08.009 https://doi.org/10.1016/j.jchf.2017.08.009 PubMed: https://pubmed.ncbi.nlm.nih.gov/29096799/ Update 2026: Dr. Jaswal is a Nuclear Medicine Resident at New York Presbyterian/ Weill Cornell Medicine. Shama Jaswal is an International Medical Graduate, currently doing research at Mallinckrodt Institute of Radiology (MIR), Saint Louis. She aims at pursuing Diagnostic Radiology residency and poses a keen interest in research alongside academics. At MIR, she has been fortunate to work on various oncology projects including the project in which they studied how the difference in fat metabolism in both sexes can affect the cancer survival and outcome, and how this study can further improve prognosis through treatment modification. Shama is both an accomplished sprinter and singer having won several national competitions in in each discipline in India. She also has a strong passion for cooking and gardening. Follow Shama Jaswal on Twitter @Jaswal_Shama All posts by Shama Jaswal Kevin M. Rice, MD is the president of Global Radiology CME and is a radiologist with Cape Radiology Group . He has held several leadership positions including Board Member and Chief of Staff at Valley Presbyterian Hospital in Los Angeles, California. Dr. Rice has made several media appearances as part of his ongoing commitment to public education. Dr. Rice's passion for state-of-the-art radiology and teaching includes acting as a guest lecturer at UCLA. In 2015, Dr. Rice and Natalie Rice founded Global Radiology CME to provide innovative radiology education at exciting international destinations, with the world's foremost authorities in their field. In 2016, Dr. Rice was nominated and became a semifinalist for a "Minnie" Award for the Most Effective Radiology Educator. He was once again a semifinalist for a "Minnie" for 2021's Most Effective Radiology Educator by AuntMinnie.com . He has continued to teach by mentoring medical students interested in radiology . Everyone he has mentored has been accepted into top programs across the country, including Harvard, UC San Diego, Northwestern, Vanderbilt, and Thomas Jefferson. Follow Dr. Rice on Twitter @KevinRiceMD All posts by Kevin M. Rice, MD
- Independent Travel from Hotel Chinzanso Tokyo to Hyatt Regency Kyoto for Imaging in Japan 2026
Quick summary From Hotel Chinzanso Tokyo to Tokyo Station: Taxi (≈30 mins) ~ ¥5,000 ($34). Tokyo to Kyoto on Shinkansen: Nozomi train, ~ 2 hr 15 min, reserved seat ~ ¥14,500 ($95) per adult. Book early as the seats may be limited. Japan Rail Pass - JRailPass.com スマートEX Carry-On Rules for Shinkansen: Suitcases under 160 cm (sum of L+W+H) can be brought onboard. Larger luggage requires reservation or baggage forwarding. Baggage forwarding (Tokyo→Kyoto): Most services are next day delivery. If available, same-day service is ~ ¥4,000 - ¥6,000 (US$28 - $44) per bag depending on size and weight. Ask concierge or Yamato/Kuroneko counter for exact quote. Kyoto Station → Hyatt Regency: Taxi ~¥2000 (US$14) / ~10 min. Full details here: 1) From Hotel Chinzanso Tokyo → Tokyo Station By taxi (fastest): ~30 minutes (typical fare about ¥5000 ) depending on traffic. Most taxi drivers in Tokyo do not speak English, so be sure to have your destination written on a card in Japanese or have the hotel doorman arrange the service. By subway / bus (cheaper): walk ~10 min to Edogawabashi Station , then subway/bus connections to central Tokyo — fares ~ ¥170–¥310 , journey 30–45 min depending on route. Tokyo Taxi 2) Tokyo Station → Kyoto Station (Shinkansen) Typical travel time: ~ 2 hours 15 minutes on Nozomi / fastest services; Hikari slightly slower. JR Central Japan Rail Pass - JRailPass.com Typical one-way fare (ordinary car): broadly ¥13,000–¥14,500 (varies by train type—Nozomi, Hikari, reserved vs non-reserved). Book early for best fares (SmartEX, JR Central online). Japan Rail Pass - JRailPass.com スマートEX Booking tips Buy tickets or reserve seats in advance via SmartEX / JR Central / station ticket counters, especially if you travel with luggage. SmartEX offers advance discounts for some fares. スマートEX JR Central Shinkansen - Bullet Train Luggage on the Shinkansen — what you need to know Standard rule: Passengers may bring luggage, but large suitcases are regulated. Luggage with total dimensions over 160 cm should be managed (either use the designated oversized baggage area or make a reservation for a seat with an oversized baggage area). Items with total dimensions up to 250 cm may be allowed as carry items (subject to limits and seat/area availability). In practice, suitcases under 160 cm total (height+width+depth) travel without special arrangements. JR Central West Japan Railway Company Practical advice If your suitcase is ≤160 cm total (e.g., 65 cm × 40 cm × 30 cm ≈ 135 cm total), you can bring it on board and stow it in the overhead or behind seats. If >160 cm (but ≤250 cm total), you must reserve a seat with an oversized-baggage area (last-row seats on some cars) — no extra ticket surcharge, but reservation is required. Reserve early. West Japan Railway Company JR Central Kyoto Station 3) Kyoto Station → Hyatt Regency Kyoto Taxi: ~10 minutes, fare around ¥2000 depending on route and traffic. Local train + short walk: ~15–20 minutes (cheaper option). See hotel access instructions on arrival. hyattregency.hotel-kyoto.com Baggage-forwarding (takkyubin) — a popular option Why use it: Send your checked suitcases from Hotel Chinzanso Tokyo to Hyatt Regency Kyoto so you travel light on the train (highly recommended). Most hotels accept and drop off for kuroneko/Yamato or Japan Post takkyubin. Typical pricing / timing (examples & guidance) Next-day delivery (standard, widely available): typical range ¥3000–¥5000 per item depending on size (smaller bags cheaper; 160-size suitcase frequently ~¥2,000–¥3,000 Tokyo→Kyoto). Same-day delivery: available from selected Yamato / Kuroneko counters on some routes — prices vary and same-day service depends on pick-up time and destination (ask the provider or hotel concierge). Expect higher fees than next-day, in the ¥4000–¥6000 per item range. Video Guide To Luggage Delivery Service How to arrange Ask Hotel Chinzanso Tokyo concierge to reserve takkyubin pickup and confirm delivery date — they will handle forms in English. Contact the concierge team directly at concierge@hotel-chinzanso.com Provide your Hyatt Regency Kyoto reservation name and arrival date (note: confirm the hotel will accept forwarded bags). Keep a copy of the takkyubin receipt and tracking number; delivery is usually next-day, but confirm if you need same-day service. Conclusion Whether you choose the Global Radiology CME seamless Kyoto Transfer and 1 Day Tour , the Kyoto Transfer and 3 Day Tour , or plan your own trip we look forward to seeing you at the Business and Wellness mini-conference in Kyoto. --Natalie and Kevin Rice
- Top 10 Reasons to Attend Imaging in Japan 2026
Don’t Miss This Unique Experience Imaging in Japan 2026 isn’t just a conference—it’s an opportunity to grow, connect, and be inspired in one of the most extraordinary countries in the world. 📅 Conference Dates: June 1–5, 2026 📍 Location: Hotel Chinzanso Tokyo + Optional Mini-Conference at Hyatt Regency Kyoto 🔗 View the full program and register now → www.globalradiologycme.com/imaging-in-japan2026 Here are the top 10 reasons you should attend Imaging in Japan 2026: 1. Leading edge, captivating Scientific Program delivered by a World-Class Faculty Led by Scientific Director Neil M. Rofsky, the 2025 program will feature topics in body imaging, cardiothoracic imaging, MSK, neuroradiology, and emergency radiology. Faculty include some of the most respected names in the field: Donald Resnick, Ella Kazerooni, Amish Doshi, and Karen Lee. 2. Tokyo is ranked one of the top cities to visit in the world Travel + Leisure ranked Tokyo #3 in their readers favorite cities in the world in 2026. Forbes ranked Tokyo #3 of Best Cities in the world to visit in 2026. 3. Interactive and engaging Lunch Round Tables Back by popular demand, our daily Round Table sessions offer a chance to discuss current imaging and leadership topics in an informal setting with the faculty. These sessions are often cited by attendees as a highlight of the educational program. 4. Hotel Chinzanso Tokyo : A Hidden Gem in the Heart of Tokyo The conference venue, Hotel Chinzanso Tokyo, is a luxurious garden retreat tucked away in central Tokyo. You’ll attend sessions surrounded by centuries-old camellia trees, koi ponds, and stone paths—an inspiring and peaceful place to learn. 5. Kyoto: A Spiritual and Cultural Retreat Don’t miss the optional Wellness and Business Mini-Conference on June 5, 2026 at the Hyatt Regency Kyoto. Explore Japan’s former imperial capital, home to over 1,600 temples, zen gardens, and world-famous sites like the Fushimi Inari Shrine and Arashiyama Bamboo Forest. 6. Cruise and Dinner on Tokyo Bay Network with fellow registrants and faculty while enjoying a spectacular evening cruise on Tokyo Bay, complete with dinner, drinks, and panoramic views of the city skyline and Rainbow Bridge. 7. Explore Tokyo’s Contrasts From the bustling energy of Shibuya Crossing to the quiet elegance of the Meiji Shrine, Tokyo is a city of contrasts. Shop in Ginza, experience a traditional tea ceremony, take a ride in a rickshaw or marvel at the views from Tokyo Skytree—all just a short ride from the conference hotel. 8. Soak in a Traditional Onsen Experience the healing waters of a Japanese onsen. Whether in Tokyo or Kyoto, you’ll find beautifully designed public baths and hotel spas that offer the perfect post-conference wind-down. 9. Savor Michelin-Starred Cuisine and Street Food Japan has more Michelin-starred restaurants than anywhere else on earth—but the culinary scene goes far beyond fine dining. Don’t leave without trying fresh sushi at Tsukiji Market, sizzling yakitori in a local izakaya, or warm taiyaki from a street vendor. 10. Ride the Shinkansen (Bullet Train) Fast, efficient, and a uniquely Japanese experience, the Shinkansen connects Tokyo and Kyoto in just over two hours. Experience an exceptional radiology education alongside a memorable cultural journey at this distinctive conference by Global Radiology CME. The Imaging in Japan 2026 event offers professional growth, networking opportunities, and wellness for radiologists worldwide in a culturally vibrant country that seamlessly integrates ancient traditions with contemporary innovations.
- Percutaneous Cholecystostomy in Pregnancy
Right upper quadrant pain in unstable patient. What procedure is indicated? • Xray of the Week Figure 1. What action should be taken for this patient with right upper quadrant pain who is also hemodynamically unstable? Figure 2. A. MRI of abdomen. The patient is pregnant (orange arrows), therefore ionizing radiation with CT scan or fluoroscopy can not be used for imaging guidance. Gallbladder with wall thickening (green arrow) and adjacent fluid (yellow arrow) indicate acute cholecystitis. A nuclear medicine hepatobiliary scan was also performed (not shown) and demonstrated no radiopharmaceutical present in the gallbladder due to cystic duct obstruction. B. Ultrasound of gallbladder used for guidance of percutaneous needle (red arrow) placement for cholecystostomy. C. Ultrasound of gallbladder demonstrating drainage catheter in the lumen (blue arrow). Discussion: Cholecystitis is the second most common surgical emergency seen in pregnancy after appendicitis. [1] Surgical intervention is typically safe for both the mother and fetus , due to improved morbidity when utilizing the laparoscopic approach rather than open cholecystectomy. [2]. In cases of high risk pregnancy or when treating an unstable peripartum patient, percutaneous cholecystostomy is an important important image-guided, minimally invasive alternative to surgical cholecystectomy [ 3-6 ]. This technique has proven effective for cases of acute cholecystitis occurring during the third trimester, allowing for management until delivery when surgery becomes safer. [7] . Percutaneous cholecystostomy is usually followed by laparoscopic cholecystectomy in the postpartum period once the patient has been stabilized [ 3,7 ]. In pregnancy, ultrasound is used for imaging guidance due to lack of ionizing radiation encountered with CT scan or fluoroscopy [5]. The transhepatic or transperitoneal insertion of an access needle is followed by gallbladder catheterization with either the Seldinger technique or a trocar system [8-11]. Figures 1 and 2 are imaging studies on a pregnant patient with acute cholecystitis and was too unstable to undergo surgery. The patient underwent a percutaneous cholecystostomy using the Seldinger technique and US guidance. After the patient delivered and was stable, the patient had a laparoscopic cholecystectomy and fully recovered. Major complications of percutaneous cholecystostomy include hemorrhage, pneumothorax, biliary leak, and peritonitis. The transhepatic approach may carry higher risk of pleural or hepatic injury, while transperitoneal avoids liver traversal but risks bowel injury. [8,9,11]. Prognosis is excellent with timely intervention, allowing safe maternal stabilization and fetal protection until definitive treatment. Key Learning Points Ultrasound-guided percutaneous cholecystostomy is a safe bridge in high-risk pregnant patients with acute cholecystitis, especially in the third trimester. Look for gallbladder distention and real-time needle/catheter placement on ultrasound to confirm access and decompression. Radiology plays a key role in avoiding radiation and guiding minimally invasive drainage—prefer transhepatic or transperitoneal based on anatomy. Prompt intervention stabilizes the patient; interval cholecystectomy is standard postpartum for definitive management. References: Angelini DJ. Obstetric triage revisited: update on non-obstetric surgical conditions in pregnancy. J Midwifery Womens Health . 2003;48(2):111-118. doi: 10.1016/s1526-9523(02)00417-8 . Knab LM, Boller AM, Mahvi DM. Cholecystitis. Surg Clin North Am . 2014;94(2):455-470. doi: 10.1016/j.suc.2014.01.005 Hojberg Y, Patel K, Shebrain S. Utilizing Percutaneous Cholecystostomy Tube as a Temporary Minimally Invasive Approach for Acute Cholecystitis during Third Trimester of a High-Risk Pregnancy. Case Rep Gastroenterol . 2022;16(1):49-54. Published 2022 Feb 14. doi: 10.1159/000522060 Baron TH , Grimm IS , Swanstrom LL . Interventional approaches to gallbladder disease . N Engl J Med . 2015 ; 373 ( 4 ): 357 – 65 . doi: 10.1056/NEJMra1411372 Moirano J, Khoury J, Yeisley C, Noor A, Voutsinas N. Interventional Radiology and Pregnancy: From Conception through Delivery and Beyond. Radiographics . 2023;43(8):e230029. doi: 10.1148/rg.230029 Rana P, Gupta P, Chaluvashetty SB, et al. Interventional radiological management of hepatobiliary disorders in pregnancy. Clin Exp Hepatol . 2020;6(3):176-184. doi: 10.5114/ceh.2020.99508 Caliskan K. The use of percutaneous cholecystostomy in the treatment of acute cholecystitis during pregnancy. Clin Exp Obstet Gynecol . 2017;44(1):11-13. https://pubmed.ncbi.nlm.nih.gov/29714857/ Ginat D and Saad W. Cholecystostomy and Transcholecystic Biliary Access. Tech Vasc Interv Radiol. 2008;11(1):2-13. doi: 10.1053/j.tvir.2008.05.002 Little MW. Percutaneous cholecystostomy: The radiologist’s role in treating acute cholecystitis. Clin Radiol. 2013;68(7): 654-660. doi: 10.1016/j.crad.2013.01.017 Venara A, Carretier V, Lebigot J, E Lermite. Technique and indications of percutaneous cholecystostomy in the management of acute cholecystitis in 2014. J Visc Surg. 2014;151(6):435-439. doi: 10.1016/j.jviscsurg.2014.06.003 Beland MD, Patel L, Ahn SH, Grand DJ. Image-Guided Cholecystostomy Tube Placement: Short- and Long-Term Outcomes of Transhepatic Versus Transperitoneal Placement. AJR Am J Roentgenol. 2019;212: 201-204. doi: 10.2214/AJR.18.19669 Kevin M. Rice, MD is the president of Global Radiology CME and is a radiologist with Cape Radiology Group . He has held several leadership positions including Board Member and Chief of Staff at Valley Presbyterian Hospital in Los Angeles, California. Dr. Rice has made several media appearances as part of his ongoing commitment to public education. Dr. Rice's passion for state of the art radiology and teaching includes acting as a guest lecturer at UCLA. In 2015, Dr. Rice and Natalie Rice founded Global Radiology CME to provide innovative radiology education at exciting international destinations, with the world's foremost authorities in their field. In 2016, Dr. Rice was nominated and became a semifinalist for a "Minnie" Award for the Most Effective Radiology Educator. He was once again a semifinalist for a "Minnie" for 2021's Most Effective Radiology Educator by AuntMinnie.com . He has continued to teach by mentoring medical students interested in radiology . Everyone who he has mentored has been accepted into top programs across the country including Harvard, UC San Diego, Northwestern, Vanderbilt, and Thomas Jefferson. Follow Dr. Rice on Twitter @KevinRiceMD All posts by Kevin M. Rice, MD
- Bilateral HeRO Grafts
52 year old Male on dialysis. What are the devices? • Xray of the Week Figure 2: Frontal chest radiograph in a dialysis patient . What are the devices? Figure 2: Frontal chest radiograph in a dialysis patient with bilateral HeRO grafts. The green arrow indicates the proximal aspect of the right-sided nitinol-reinforced venous component entering the central circulation via the internal jugular vein. The red arrow highlights the left-sided system entering via the subclavian vein. The yellow arrow identifies the convergence of the dual mesh-like tubes as they cross the cavoatrial junction. Figure 3: Anatomical diagram of a right-sided HeRO graft implantation. The arterial graft component (with cannulation area indicated) is anastomosed to the brachial artery and tunneled subcutaneously in the upper arm, connecting via the titanium connector to the venous outflow component. The venous outflow component bypasses central venous stenosis, entering the central circulation (typically via the internal jugular vein) and terminating in the right atrium for continuous outflow. Radiologic Evaluation and Management of Bilateral HeRO Grafts in Access-Challenged Patients Discussion The clinical course of patients with end-stage kidney disease (ESKD) is often limited by "vascular access exhaustion." Central venous stenosis (CVS) or occlusion (CVO) occurs in up to 40% of patients who have had prior central venous catheters, leading to venous hypertension and the failure of standard arteriovenous fistulas or grafts.[3] For these access-challenged patients, the Hemodialysis Reliable Outflow (HeRO) graft provides a critical salvage option by bypassing central obstructions to drain blood directly into the right atrium.[1][2] System Components and Radiographic Appearance The HeRO system is a hybrid device consisting of three subcutaneous parts that a general radiologist must recognize on routine imaging (see Figure 1 for anatomical overview): Arterial Graft Component: A 6-mm inner diameter expanded polytetrafluoroethylene (ePTFE) graft. On radiographs, it is poorly radiopaque, but it features distinctive PTFE beading near its distal end to provide kink resistance.[2] Titanium Connector: This is a highly radiopaque, tapered junctional device typically located in the deltopectoral groove. It joins the arterial graft to the venous outflow component.[2] Venous Outflow Component (VOC): This is a 19-French radiopaque silicone tube reinforced with a 48-braid nitinol mesh. It produces a dense, mesh-like "necklace" appearance on chest radiographs, which is the hallmark of the device.[2] Imaging Findings In the provided frontal chest radiograph, a rare bilateral HeRO graft configuration is visualized. This setup is typically reserved for patients with total, bilateral central venous occlusions who have exhausted all other upper-extremity options.[3][8] The green arrow identifies the right-sided system, where the reinforced nitinol component enters the central circulation via the right internal jugular vein. The red arrow points to the contralateral left-sided system entering via the left subclavian vein. The yellow arrow points to the central convergence of the two nitinol-reinforced components at the cavoatrial junction. The distal tips of these components should ideally reside in the mid-to-upper right atrium to ensure optimal flow and minimize the risk of thrombus formation.[2] See Figure 3 for a labeled anatomical diagram illustrating the right-sided access, including the cannulation area on the arterial graft component, the venous outflow path, and overall subcutaneous routing. While radiographs are used for routine surveillance, contrast-enhanced CT venography (CTV) or ferumoxytol-enhanced MR venography (MRV) is the gold standard for preoperative mapping of occlusions and collateral pathways.[9] Ultrasound is primarily used to evaluate the superficial arterial graft segment for patency and volume flow (typically 800–1,000 mL/min).[2] Differential Diagnosis Radiologists must differentiate the HeRO system from other common thoracic devices: Standard Tunneled Dialysis Catheters (TDCs): These are non-reinforced, less radiopaque, and lack the titanium connector and nitinol mesh "necklace" appearance.[2] Central Venous Stents: While mesh-like, stents are entirely intravascular and are not connected to a subcutaneous arterial graft.[3] Pacemaker and AICD Leads: These are much thinner and terminate in specific cardiac chambers (e.g., right ventricular apex). They do not feature the wide-caliber mesh seen in HeRO components.[2] Management and Prognosis The HeRO graft offers a major clinical advantage over standard catheters by being entirely subcutaneous, which reduces the risk of bacteremia by approximately 69%.[5] Patency rates are comparable to conventional ePTFE grafts, with secondary patency—the ability to maintain function through interventions—reported at 80% to 90% at one year.[4][6] However, the device requires frequent maintenance. On average, patients require 1.5 to 2.2 radiologic interventions per year to treat complications such as graft thrombosis or component migration.[7] Migration of the nitinol component (either retracting into the SVC or advancing into the IVC) is a serious complication that can lead to device dysfunction or atrial injury.[7] Evolving therapies for underlying AVF/graft dysfunction (e.g., drug-coated balloons, endovascular AVF creation) may complement HeRO in select cases.[11] Key Learning Points Recognition: Identify the HeRO graft by its dense nitinol-braid "necklace" appearance and the highly radiopaque titanium connector in the deltopectoral groove.[2] Bilateral Configuration: Bilateral grafts represent a salvage strategy for patients with total thoracic central vein obstruction (TCVO).[3][8] Clinical Value: The subcutaneous design significantly lowers infection rates compared to tunneled catheters.[5] Radiologist's Role: General radiologists are often the first to identify device complications like kinking at the connector or tip migration on routine chest films.[7] Advanced Imaging: Ferumoxytol-enhanced MRV is a safe alternative for venous mapping in patients with severe renal impairment.[9] References: Katzman HE, McLafferty RB, Ross JR, Glickman MH, Peden EK, Lawson JH. Initial experience and outcome of a new hemodialysis access device for catheter-dependent patients. J Vasc Surg . 2009;50(3):600-607. doi: 10.1016/j.jvs.2009.04.014 Tabriz DM, Arslan B. HeRO Graft: Indications, Technique, Outcomes, and Secondary Intervention. Semin Intervent Radiol . 2022;39(1):82-89. doi: 10.1055/s-0042-1742391 Fitzgerald KM, Newell KR, Shin DS, et al. Thoracic Central Vein Obstruction: Endovascular Recanalization and Reconstruction. Radiographics . 2024;44(1):e240178. doi: 10.1148/rg.240178 Gage SM, Katzman HE, Ross JR, et al. Multi-center experience of 164 consecutive Hemodialysis Reliable Outflow graft implants for hemodialysis treatment. Eur J Vasc Endovasc Surg . 2012;44(1):93-99. doi: 10.1016/j.ejvs.2012.04.011 Kim Y, Cui CL, Eze AN, et al. Perioperative and long-term outcomes after Hemodialysis Reliable Outflow (HeRO) graft surgery. J Vasc Surg . 2025;82(4):1458-1466.e1. doi: 10.1016/j.jvs.2025.05.042 Gebhard TA, Bryant JA, Adam Grezaffi J, et al. Percutaneous interventions on the hemodialysis reliable outflow vascular access device. J Vasc Interv Radiol . 2013;24(4):543-549. doi: 10.1016/j.jvir.2012.12.027 Cline BC, Zuchowski A, Gage SM, et al. Hemodialysis Reliable Outflow (HeRO) graft creation in upper extremities abandoned due to stent obstruction via recanalization and HeRO outflow component insertion across stent interstices. J Vasc Access . 2021;24(5):989-993. Published 2021 Dec 10. doi: 10.1177/11297298211048061 Medjahed O, Jha AK, Ahmed A, Shaat A. Point-of-Care Ultrasound in the Diagnosis of Pulmonary Embolism With Right Heart Strain: A Case Report of Dialysis Line-Related Complications. Cureus . 2025;17(11):e96273. Published 2025 Nov 7. doi: 10.7759/cureus.96273 Takahashi EA, Kilari S, Misra S. Novel Clinical Therapies and Technologies in Dialysis Vascular Access. Kidney360 . 2021;2(8):1373-1379. Published 2021 Jun 10. doi: 10.34067/KID.0002962021 Lok CE, Huber TS, Lee T, et al. KDOQI Clinical Practice Guideline for Vascular Access: 2019 Update. Am J Kidney Dis . 2020;75(4 Suppl 2):S1-S164. doi: 10.1053/j.ajkd.2019.12.001 Ekanem AA, Eze AN, Eze AN, Cui CL, Kim Y, Southerland KW. A rare case of hemorrhage from spontaneous disconnection of super-HeRO adapter and early-cannulation graft: A case report. J Vasc Access . 2026;27(1):361-365. doi: 10.1177/11297298251326970 Kevin M. Rice, MD is the president of Global Radiology CME and is a radiologist with Cape Radiology Group . He has held several leadership positions including Board Member and Chief of Staff at Valley Presbyterian Hospital in Los Angeles, California. Dr. Rice has made several media appearances as part of his ongoing commitment to public education. Dr. Rice's passion for state-of-the-art radiology and teaching includes acting as a guest lecturer at UCLA. In 2015, Dr. Rice and Natalie Rice founded Global Radiology CME to provide innovative radiology education at exciting international destinations, with the world's foremost authorities in their field. In 2016, Dr. Rice was nominated and became a semifinalist for a "Minnie" Award for the Most Effective Radiology Educator. He was once again a semifinalist for a "Minnie" for 2021's Most Effective Radiology Educator by AuntMinnie.com . He has continued to teach by mentoring medical students interested in radiology . Everyone he has mentored has been accepted into top programs across the country, including Harvard, UC San Diego, Northwestern, Vanderbilt, and Thomas Jefferson. Follow Dr. Rice on Twitter @KevinRiceMD All posts by Kevin M. Rice, MD
- Brachydactyly Type A3
50-year-old female presenting with short 5th finger . What is the diagnosis? • Xray of the Week Figure 1. Frontal bilateral hand X-ray. What is the diagnosis? Figure 2. Frontal bilateral hand X-ray: Severe shortening of the middle phalanx of the fifth digit (red arrows). The phalanx measures less than 50% of the length of the adjacent fourth middle phalanx, satisfying the Hertzog criteria for Brachydactyly Type A3. A mild radial clinodactyly is present, secondary to the wedge-shaped morphology of the middle phalanx. Discussion Brachydactyly (BD) refers to disproportionately short fingers and toes, classified by the Julia Bell system into five primary types (A–E) based on anatomical involvement. Type A3 (BDA3), also known as brachymesophalangy V, is the most common isolated hand anomaly.[1] Imaging Findings and Analysis Radiographs of this 50-year-old female (Fig 1, 2) reveal isolated, bilateral shortening of the middle phalanx of the fifth digit. The definitive diagnosis is established using the Hertzog criterion, which mandates that the longitudinal length of the fifth middle phalanx must be less than 50% of the longitudinal length of the fourth middle phalanx.[1] Morphologically, the affected phalanx exhibits a characteristic rhomboid or wedge-shaped configuration. The radial side of the phalanx is significantly shorter than the ulnar side, creating a slanted distal articular surface that drives radial clinodactyly (radial deflection of the distal phalanx toward the fourth digit). Pathophysiological and Genetic Mechanisms BDA3 is an autosomal dominant disorder arising from disrupted cartilage ossification during early embryonic development (blastogenesis), with some cases mapped to the chromosome 13q33 region.[1] Epidemiology and Biocultural Insights The prevalence of BDA3 varies significantly by ethnicity; it is a common variant in Asian populations, occurring in 21% to 25.6% of Japanese children, but remains rare in populations of European or African descent (<2%).[7][8] Growth and Development Although BDA3 is more frequently identified in children with short stature, it does not negatively impact the efficacy of growth hormone therapy, indicating that the local physis disruption does not represent global resistance to growth signals.[4] Syndromic Associations and Differentials Radiologists should view BDA3 as a clinical marker for broader genetic conditions, most notably Down syndrome (present in ~60%) and Turner syndrome.[9] Differential considerations include Kirner deformity (palmo-radial bowing of the distal phalanx shaft) and Camptodactyly (a soft-tissue flexion contracture of the PIP joint).[5] Genetic Counseling Inheritance is typically autosomal dominant with a 50% recurrence risk for offspring; however, for isolated cases, patients should be reassured that BDA3 is a benign anatomical variant.[1] Management and Prognosis Isolated BDA3 is almost universally asymptomatic and requires no medical or surgical intervention. The functional prognosis is excellent, as the shortening of the little finger does not typically affect manual dexterity or grip strength.[5] Corrective surgical procedures, such as osteotomy, are extremely rare and indicated only for severe clinodactyly that causes functional impairment or significant cosmetic distress.[1] Key Learning Points Diagnostic Standard : BDA3 is confirmed when the fifth middle phalanx length is <50% of the fourth (Hertzog criterion) .[1] Radiographic Features : Look for a wedge-shaped middle phalanx and associated radial clinodactyly. Population Variant : High prevalence in Japanese and other Asian populations should be noted as a common anatomical variation .[7][8] Clinical Marker : Identification of BDA3 warrants screening for Down and Turner syndromes, particularly if associated with metacarpal shortening .[9] Management : Isolated BDA3 is benign; functional impairment is rare, though corrective options exist for severe cases .[5] References Temtamy SA, Aglan MS. Brachydactyly. Orphanet J Rare Dis . 2008;3:15. doi: 10.1186/1750-1172-3-15 Garn SM, Hertzog KP, Poznanski AK, Nagy JM. Metacarpophalangeal length in the evaluation of skeletal malformation. Radiology . 1972;105(2):375-381. doi: 10.1148/105.2.375 Wu H, Wu H, Li Y, Li H. Brachydactyly Type A3 Is More Commonly Seen in Children With Short Stature But Does Not Affect Their Height Improvement by Growth Hormone Therapy. Front Endocrinol (Lausanne) . 2022;13:824315. doi: 10.3389/fendo.2022.824315 Nguyen ML, Jones N. Undergrowth: brachydactyly. Hand Clin . 2009;25(2):247-255. doi: 10.1016/j.hcl.2009.02.003 Everman DB. The brachydactylies. In: Stevenson RE, Hall JG, eds. Human Malformations and Related Anomalies . 2nd ed. Oxford University Press; 2006:968-983. Zhang W, Li K, Zhang Q, et al. Epidemiology of brachydactyly type A3 in China: a nationwide multicentre population-based study among children aged 3–17 years. BMJ Open . 2025;15(11):e099166. doi: 10.1136/bmjopen-2025-099166 Wu HH, Zhang YQ, Yu CD, et al. Brachydactyly type A3 may be associated with shorter stature: An observation from a Chinese pediatric sample. PLoS One . 2025;20(11):e0336913. doi: 10.1371/journal.pone.0336913 Kang MJ, Kanakatti Shankar R, Jee YH. Phalangeal bone growth and implications in Turner syndrome. Front Endocrinol (Lausanne) . 2026;16:1735962. doi: 10.3389/fendo.2025.1735962 . Castriota-Scanderbeg A, Dallapiccola B. Abnormal Skeletal Phenotypes: From Sameness to Differential Diagnosis . Springer; 2006. doi: 10.1007/3-540-30361-8 Cleveland Clinic. Brachydactyly. Accessed January 20, 2026. https://my.clevelandclinic.org/health/diseases/24081-brachydactyly Kevin M. Rice, MD is the president of Global Radiology CME and is a radiologist with Cape Radiology Group . He has held several leadership positions including Board Member and Chief of Staff at Valley Presbyterian Hospital in Los Angeles, California. Dr. Rice has made several media appearances as part of his ongoing commitment to public education. Dr. Rice's passion for state-of-the-art radiology and teaching includes acting as a guest lecturer at UCLA. In 2015, Dr. Rice and Natalie Rice founded Global Radiology CME to provide innovative radiology education at exciting international destinations, with the world's foremost authorities in their field. In 2016, Dr. Rice was nominated and became a semifinalist for a "Minnie" Award for the Most Effective Radiology Educator. He was once again a semifinalist for a "Minnie" for 2021's Most Effective Radiology Educator by AuntMinnie.com . He has continued to teach by mentoring medical students interested in radiology . Everyone he has mentored has been accepted into top programs across the country, including Harvard, UC San Diego, Northwestern, Vanderbilt, and Thomas Jefferson. Follow Dr. Rice on Twitter @KevinRiceMD All posts by Kevin M. Rice, MD
- Arachnoiditis Ossificans
An 74-year-old male with chronic low back pain and progressive lower extremity weakness. What is the diagnosis? • Xray of the Week Figure 1. Non-contrast CT of the lumbar spine. What is the diagnosis? Figure 2. Non-contrast CT of the lumbar spine (axial, sagittal, and coronal reconstructions): Central pattern of arachnoiditis ossificans demonstrating dagger-like ossification within the central spinal canal at the L5 level (red arrows). The linear, hyperdense bony bar courses longitudinally along the central canal, causing focal narrowing and potential cauda equina compression. No peripheral or circumferential involvement is seen, consistent with the central ossification pattern. Diagnosis Arachnoiditis ossificans. Discussion Arachnoiditis ossificans is a rare end-stage manifestation of chronic adhesive arachnoiditis, characterized by ossification of the arachnoid membrane within the spinal canal.[1][2] It typically occurs in the thoracolumbar region and is associated with prior spinal surgery, trauma, infection, subarachnoid hemorrhage, or oil-based myelography.[3][4] Patients often present with progressive myelopathy or radiculopathy due to nerve root compression or tethering.[5] The condition represents metaplastic ossification of inflamed arachnoid tissue, leading to intrathecal calcified or ossified plaques that can encase the spinal cord or cauda equina.[6] Imaging Findings Computed tomography (CT) is the modality of choice for diagnosing arachnoiditis ossificans due to its superior depiction of ossified structures, with sensitivity for detecting intrathecal ossification approaching 100% in symptomatic cases.[2][7] On CT, hyperdense bone attenuation plaques are seen within the thecal sac, with patterns varying by type of ossification [2]: Central pattern : Linear or dagger-like hyperdense ossification centered within the spinal canal, often appearing as a solitary midline bony bar or rod on axial views. (Figs. 1,2) Nerve root encasing pattern : Tubular or circumferential hyperdense ossifications surrounding individual or clumped nerve roots (cauda equina), with roots appearing embedded or passing through the ossified tissue. Weblike pattern : Interlacing, mesh-like hyperdense strands or trabeculae filling or crisscrossing the thecal sac, creating a reticular appearance between nerve roots. Peripheral pattern : Rim-like or discontinuous hyperdense plaques along the inner walls of the thecal sac, often circumferential or partially encircling, potentially narrowing the canal without central involvement. These patterns frequently coexist, leading to thecal sac deformity, nerve root clumping, and varying degrees of central canal stenosis. Multiplanar reconstructions (sagittal/coronal) help confirm the distribution and extent. Patterns of intrathecal ossification can guide prognosis and surgical planning. Management and Prognosis Management is primarily conservative, focusing on pain control and physical therapy, as surgical decompression carries high risks of reossification or worsening symptoms.[3][5] Laminectomy with duraplasty may be considered for severe cord compression, with variable success rates (30-50% improvement in symptoms).[4] Prognosis is guarded, with many patients experiencing progressive disability; early detection via imaging can facilitate timely intervention to prevent irreversible myelopathy.[6] Key Learning Points CT is essential for identifying hyperdense intrathecal ossifications in arachnoiditis ossificans, outperforming MRI for bony detail. Look for circumferential or patterned ossified plaques encasing nerve roots on multiplanar reconstructions to differentiate from mimics like dural calcifications. Radiology plays a critical role in classifying ossification patterns, which influence surgical feasibility and prognosis. Prompt recognition on CT can guide multidisciplinary management and prevent complications like syringomyelia—always correlate with clinical history of prior spinal insult. References Frizzell B, Kaplan P, Dussault R, Sevick R. Arachnoiditis ossificans: MR imaging features in five patients. AJR Am J Roentgenol . 2001;177(2):461-464. doi: 10.2214/ajr.177.2.1770461 Thejeel B, Greditzer-Sobeck C, Ciacci J, Siddiqi I. Patterns of intrathecal ossification in arachnoiditis ossificans: a retrospective case series. AJNR Am J Neuroradiol . 2023;44(2):228-234. doi: 10.3174/ajnr.A7764 Donalisio M, Cadosch D. Arachnoiditis ossificans. Skeletal Radiol . 2024;53(5):1019-1021. doi: 10.1007/s00256-023-04465-7 Junewick JJ. Clinical image. Arachnoiditis ossificans in a pediatric patient. Pediatr Radiol . 2010;40(2):228. doi: 10.1007/s00247-009-1350-2 Jaspan T, Preston BJ, Mulholland RC, Webb JK. The CT appearances of arachnoiditis ossificans. Spine (Phila Pa 1976) . 1990;15(2):148-151. doi: 10.1097/00007632-199002000-00022 Chan CC, Lau PY, Sun LK, Lo SS. Arachnoiditis ossificans. Hong Kong Med J. 2009;15(2):146-148. https://pubmed.ncbi.nlm.nih.gov/19342743/ Kumaran SP, Gupta K, Maddali A, Viswamitra S. Post traumatic arachnoiditis ossificans. J Emerg Trauma Shock . 2012;5(3):250-252. doi: 10.4103/0974-2700.99701 Sefczek RJ, Deeb ZL. Case report: computed tomography findings in spinal arachnoiditis ossificans. J Comput Tomogr . 1983;7(3):315-318. doi: 10.1016/0149-936x(83)90099-1 Kevin M. Rice, MD is the president of Global Radiology CME and is a radiologist with Cape Radiology Group . He has held several leadership positions including Board Member and Chief of Staff at Valley Presbyterian Hospital in Los Angeles, California. Dr. Rice has made several media appearances as part of his ongoing commitment to public education. Dr. Rice's passion for state-of-the-art radiology and teaching includes acting as a guest lecturer at UCLA. In 2015, Dr. Rice and Natalie Rice founded Global Radiology CME to provide innovative radiology education at exciting international destinations, with the world's foremost authorities in their field. In 2016, Dr. Rice was nominated and became a semifinalist for a "Minnie" Award for the Most Effective Radiology Educator. He was once again a semifinalist for a "Minnie" for 2021's Most Effective Radiology Educator by AuntMinnie.com . He has continued to teach by mentoring medical students interested in radiology . Everyone he has mentored has been accepted into top programs across the country, including Harvard, UC San Diego, Northwestern, Vanderbilt, and Thomas Jefferson. Follow Dr. Rice on Twitter @KevinRiceMD All posts by Kevin M. Rice, MD
- Hyphema with Vitreous Hemorrhage
Pt fell and his eye hit a metal object. What is the diagnosis? • Xray of the Week Figure 1. Non-contrast CT orbits – axial and sagittal images. What is the diagnosis? Figure 2. Non-contrast CT orbits – axial and sagittal images. Hyperdense material consistent with blood fills the anterior chamber of the left eye (red arrows), indicating hyphema. Hyperdense material is also present in the vitreous chamber posteriorly (yellow arrows), consistent with acute vitreous hemorrhage. The right globe shows a normal lens separating anterior and posterior chambers (green arrows) for comparison. Diagnosis Traumatic hyphema with concurrent vitreous hemorrhage in the left eye. Discussion Hyphema is hemorrhage into the anterior chamber, most commonly from blunt or penetrating ocular trauma, but also possible from surgery, coagulopathy, or spontaneous causes (e.g., iris neovascularization).[1][2] Vitreous hemorrhage involves bleeding into the vitreous cavity, often linked to trauma, proliferative diabetic retinopathy, retinal tears, or vascular occlusions.[3] In this traumatic case, both findings coexist due to shearing forces disrupting iris/ciliary body vessels (hyphema) and posterior structures (vitreous hemorrhage). Hyphemas are graded clinically by the volume of layered blood in the anterior chamber (Table below). Grading helps predict complications like rebleeding (highest in grades III–IV) and secondary glaucoma.[4][5][6] Hyphema Grade Approximate Volume of Blood in the Anterior Chamber Risk of IOP elevation/ secondary glaucoma Grade 0: Microhyphema <1% (slit-lamp only) <10% Grade I <33% ~10% Grade II 33–50% ~10% Grade III >50% but not full ~25% Grade IV 100% >50% "Eight ball" hyphema 100% & dark color due to poor circulation 100% Figure 3. Traumatic hyphema c linical image: Note the layering blood in the anterior chamber in this patient following blunt eye trauma. Contributor : Jesse Vislisel, MD - EyeRounds.org The University of Iowa. Creative Commons 3.0 https://webeye.ophth.uiowa.edu/eyeforum/atlas/pages/Hyphema/index.htm#gsc.tab=0 Imaging Findings Non-contrast orbital CT is the preferred initial imaging for acute ocular trauma when direct exam is limited by swelling, pain, or suspected open globe.[7][8] Hyphema appears as hyperdense (blood attenuation ~40–70 HU) material layering or filling the anterior chamber, anterior to the lens.[7] Vitreous hemorrhage shows increased attenuation in the posterior segment, often homogeneous acutely or heterogeneous if clotted/organized.[3][9] Reported sensitivity for detecting hyphema on CT is ~77%, specificity ~88%.[7] CT excels at identifying associated injuries (e.g., lens dislocation, globe rupture, foreign bodies, orbital fractures) and is safer than ultrasound if open globe is suspected (to avoid pressure on the eye).[8] Ultrasound is superior for posterior segment details (e.g., retinal detachment ) when media opacity from hemorrhage limits fundus view, but CT is first-line for trauma screening.[3][9] Management & Prognosis Management is primarily ophthalmology-directed. For traumatic hyphema: conservative measures include protective eye shielding, head elevation (30–45°), limited activity/bed rest, avoiding aspirin/NSAIDs/anticoagulants, and serial IOP monitoring to prevent rebleeding (peak risk days 3–5) or secondary glaucoma.[4][5][6][10] Topical corticosteroids reduce inflammation; cycloplegics (e.g., atropine) relieve ciliary spasm/pain. Antifibrinolytics (e.g., tranexamic acid or aminocaproic acid) may reduce rebleeding risk but lack strong evidence for improving final visual acuity and can prolong clot clearance—use is controversial per recent reviews.[10] There are ongoing debates regarding optimal approaches due to the absence of standardized guidelines, including medical agents, surgical techniques, and special situations (e.g., sickle-cell disease).[8] Surgical evacuation (e.g., anterior chamber washout) is indicated for large/persistent hyphema causing corneal blood staining, uncontrolled IOP, or active rebleeding. Vitreous hemorrhage often resolves spontaneously but may require vitrectomy if non-clearing or associated with retinal pathology.[3] Prognosis is generally good for isolated/low-grade cases (most resolve within days to weeks), but worse with higher grades, rebleeding, secondary glaucoma, sickle cell trait/disease, or extensive posterior involvement (e.g., retinal detachment). Approximately 5% of traumatic hyphemas require surgery.[4][5] Key Learning Points Look for hyperdense anterior chamber fluid on non-contrast CT to diagnose hyphema in trauma. Vitreous hyperdensity indicates posterior hemorrhage; always assess for associated globe/orbital injuries. CT is valuable when clinical exam is limited; ultrasound complements for posterior evaluation. Grade hyphema clinically to guide prognosis and intervention—prompt ophthalmology consultation is essential to prevent vision loss. References Sung EK, Nadgir RN, Fujita A, et al. Injuries of the globe: what can the radiologist offer? Radiographics . 2014;34(3):764-776. doi: 10.1148/rg.343135120 Hallinan JTPD, Pillay P, Koh L, Goh K, Yu W. Eye globe abnormalities on MR and CT in adults: an anatomical approach. Korean J Radiol . 2016;17(5):664-673. doi: 10.3348/kjr.2016.17.5.664 Spraul CW, Grossniklaus HE. Vitreous hemorrhage. Surv Ophthalmol . 1997;42(1):3-39. doi: 10.1016/S0039-6257(97)84041-6 Hartness E, Garza Reyes A, Yu C, Sears N. Hyphema: diagnosis and management. EyeRounds.org . February 20, 2024. Accessed January 14, 2026. https://eyerounds.org/cases/345-hyphema.htm Chen EJ, Fasiuddin A. Management of traumatic hyphema and prevention of its complications. Cureus . 2021;13(6):e15771. doi: 10.7759/cureus.15771 Miller SC, Meeralakshmi P, Fliotsos MJ, et al. Global current practice patterns for the management of hyphema. Clin Ophthalmol . 2022;16:3135-3144. doi: 10.2147/OPTH.S372273 Chazen JL, El-Sayed IH, Vance S, et al. CT in the evaluation of acute injuries of the anterior eye segment. AJR Am J Roentgenol . 2018;210(3):W107-W113. doi: 10.2214/AJR.17.18279 Bansal S, Gunasekaran PK, Azad S, Agrawal R. Controversies in the pathophysiology and management of hyphema. Surv Ophthalmol . 2016;61(3):297-308. doi: 10.1016/j.survophthal.2015.11.005 Rabinowitz R, Yagev R, Shoham A, Lifshitz T. Comparison between clinical and ultrasound findings in patients with vitreous hemorrhage. Eye (Lond) . 2004;18(3):253-256. doi: 10.1038/sj.eye.6700632 Woreta FA, Lindsley KB, Gharaibeh A, et al. Medical interventions for traumatic hyphema. Cochrane Database Syst Rev . 2023;2023(3):CD005431. doi: 10.1002/14651858.CD005431.pub5 Kevin M. Rice, MD is the president of Global Radiology CME and is a radiologist with Cape Radiology Group . He has held several leadership positions including Board Member and Chief of Staff at Valley Presbyterian Hospital in Los Angeles, California. Dr. Rice has made several media appearances as part of his ongoing commitment to public education. Dr. Rice's passion for state of the art radiology and teaching includes acting as a guest lecturer at UCLA. In 2015, Dr. Rice and Natalie Rice founded Global Radiology CME to provide innovative radiology education at exciting international destinations, with the world's foremost authorities in their field. In 2016, Dr. Rice was nominated and became a semifinalist for a "Minnie" Award for the Most Effective Radiology Educator. He was once again a semifinalist for a "Minnie" for 2021's Most Effective Radiology Educator by AuntMinnie.com . He has continued to teach by mentoring medical students interested in radiology . Everyone who he has mentored has been accepted into top programs across the country including Harvard, UC San Diego, Northwestern, Vanderbilt, and Thomas Jefferson. Follow Dr. Rice on Twitter @KevinRiceMD All posts by Kevin M. Rice, MD
- Active Bleeding in the Ascending Colon
An 83-year-old male with rectal bleeding. What is the diagnosis? • Xray of the Week Figure 1. CTA abdomen/pelvis. What is the diagnosis? Figure 2. CT angiogram: A. Non-contrast images are normal (yellow arrow) . B. Arterial phase image: Hyperdense extravasation of contrast within the lumen of the ascending colon (blue arrow), indicating active bleeding. C and D. Note the dependent layering of contrast on portal venous phase images, confirming active extravasation. Diagnosis Active gastrointestinal bleeding in the ascending colon. Discussion Gastrointestinal (GI) bleeding is a common clinical problem, particularly in the elderly, where lower GI sources predominate. Common etiologies include diverticulosis, angiodysplasia, neoplasms, ischemia, and inflammatory conditions.[1][2] In this case, the ascending colon involvement suggests a right-sided colonic source, often angiodysplasia or diverticular bleed, which can present with painless hematochezia.[3] Active bleeding is defined as extravasation of contrast into the bowel lumen on imaging, distinguishing it from pseudo-extravasation mimics like hyperdense pills or fecal material.[4] Imaging Findings Multiphase computed tomography angiography (CTA) is the preferred initial imaging for hemodynamically stable patients with suspected acute GI bleeding, offering high sensitivity (85-89%) and specificity (85-95%) for detecting active extravasation.[5][6] It is rapid, widely available, and guides subsequent interventions like endoscopy or embolization.[7] On non-contrast phase: No extravasation; may show hyperdense sentinel clot (40-70 HU) or bowel wall abnormalities. On arterial phase: Active extravasation appears as hyperdense focus (>90 HU) within the lumen, often eccentric or jet-like, increasing in size or density compared to non-contrast. On portal venous/delayed phase: Extravasation persists or enlarges, confirming active bleed; helps differentiate from venous sources. Reported detection rates for active bleeding exceed 0.3-0.5 mL/min, superior to nuclear medicine in many settings.[8] CTA also identifies structural causes (e.g., diverticula, tumors) even without active bleeding.[4] Management and Prognosis Management begins with hemodynamic resuscitation. For active lower GI bleeding confirmed on CTA, options include urgent colonoscopy (if stable), interventional radiology embolization (targeted to the bleeding vessel), or surgery for refractory cases.[1][3][7] Embolization success rates are 70-90%, with low rebleeding if superselective.[2] Prognosis depends on bleed severity, comorbidities, and etiology; elderly patients have higher mortality (10-20% for severe bleeds). Rebleeding risk is 10-25% for diverticular sources.[6] Key Learning Points Multiphase CTA is first-line for localizing active GI bleeding in stable patients, with high accuracy for extravasation. Look for hyperdense intraluminal contrast on post-contrast phases that is absent on non-contrast to confirm active bleed. CTA guides therapy by identifying the site and potential etiology; always assess for mimics like hyperdense foreign material. Prompt radiology interpretation is crucial to expedite embolization or endoscopy and improve outcomes. References Artigas JM, Martí M, Soto JA, Esteban H, Pinilla I, Guillén E. Multidetector CT angiography for acute gastrointestinal bleeding: technique and findings. Radiographics . 2013;33(5):1453-1470. doi: 10.1148/rg.335125072 Wells ML, Hansel SL, Bruining DH, et al. CT for evaluation of acute gastrointestinal bleeding. Radiographics . 2018;38(4):1089-1107. doi: 10.1148/rg.2018170138 Wortman JR, Landman W, Fulwadhva UP, Viscomi SG, Sodickson AD. CT angiography for acute gastrointestinal bleeding: what the radiologist needs to know. Br J Radiol . 2017;90(1075):20170076. doi: 10.1259/bjr.20170076 Di Serafino M, Iacobellis F, Schillirò ML, et al. The role of CT-angiography in the acute gastrointestinal bleeding: a pictorial essay of active and obscure findings. Tomography . 2022;8(5):2369-2402. doi: 10.3390/tomography8050198 Wu LM, Xu JR, Yin Y, Qu XH. Usefulness of CT angiography in diagnosing acute gastrointestinal bleeding: a meta-analysis. World J Gastroenterol . 2010;16(31):3957-3963. doi: 10.3748/wjg.v16.i31.3957 García-Blázquez V, Vicente-Bártulos A, Olavarria-Delgado A, Plana MN, van der Winden D, Zamora J. Accuracy of CT angiography in the diagnosis of acute gastrointestinal bleeding: systematic review and meta-analysis. Eur Radiol . 2013;23(5):1181-1190. doi: 10.1007/s00330-012-2721-x Kim BS, Li BT, Engel A, Samra JS, Clarke S, Norton ID, Li AE. Diagnosis of gastrointestinal bleeding: a practical guide for clinicians. World J Gastrointest Pathophysiol . 2014;5(4):467-478. doi: 10.4291/wjgp.v5.i4.467 Parekh PJ, Buerlein RC, Shams R, Vingan H, Johnson DA. Evaluation of gastrointestinal bleeding: update of current radiologic strategies. World J Gastrointest Pharmacol Ther. 2014;5(4):200-208. doi: 10.4292/wjgpt.v5.i4.200 Kevin M. Rice, MD is the president of Global Radiology CME and is a radiologist with Cape Radiology Group . He has held several leadership positions including Board Member and Chief of Staff at Valley Presbyterian Hospital in Los Angeles, California. Dr. Rice has made several media appearances as part of his ongoing commitment to public education. Dr. Rice's passion for state-of-the-art radiology and teaching includes acting as a guest lecturer at UCLA. In 2015, Dr. Rice and Natalie Rice founded Global Radiology CME to provide innovative radiology education at exciting international destinations, with the world's foremost authorities in their field. In 2016, Dr. Rice was nominated and became a semifinalist for a "Minnie" Award for the Most Effective Radiology Educator. He was once again a semifinalist for a "Minnie" for 2021's Most Effective Radiology Educator by AuntMinnie.com . He has continued to teach by mentoring medical students interested in radiology . Everyone he has mentored has been accepted into top programs across the country, including Harvard, UC San Diego, Northwestern, Vanderbilt, and Thomas Jefferson. Follow Dr. Rice on Twitter @KevinRiceMD All posts by Kevin M. Rice, MD
- Takayasu Arteritis
25-Year-Old Female with Abdominal Pain and Weight Loss: Diagnosis? • Xray of the Week Figure 1. 25-Year-Old Female with Abdominal Pain and Weight Loss: Diagnosis? Figure 2. CTA through the thoracic and abdominal aorta. A. Axial image through the descending thoracic aorta demonstrates concentric mural thickening and mild stenosis (yellow arrow). B. Axial image through the infra-renal abdominal aorta demonstrates mild concentric mural thickening and severe stenosis (red arrow). C. CTA 3D Image demonstrates severe stenosis of the infra-renal abdominal aorta and very severe stenosis of the origin of the common iliac arteries (green arrow). Takayasu Arteritis Epidemiology Takayasu arteritis, named after Mikito Takayasu, is a rare large-vessel vasculitis. This condition is also known as pulseless disease. The global prevalence ranges from 3.2 to 40 cases per million, with an annual incidence of 0.4 to 2.6 per million, depending on geographic location. It predominantly affects young women, with a female-to-male ratio of approximately 8 to 9:1. Clinical Findings Patients with Takayasu arteritis often present with constitutional symptoms. These may include weight loss, fever, and malaise, which develop gradually. As the disease progresses, vascular symptoms can emerge. Abdominal pain may indicate mesenteric ischemia due to stenosis in the abdominal aorta or its branches. A physical examination may reveal diminished peripheral pulses, differing blood pressures between arms, bruits over major arteries, and elevated inflammatory markers such as ESR and CRP. Pathology Histologically, Takayasu arteritis is characterized by granulomatous inflammation of the adventitia and media. This includes giant cells, lymphocytic infiltration, and intimal hyperplasia. Over time, progressive fibrosis leads to concentric wall thickening, which can result in stenosis, occlusion, or aneurysmal changes. Classification The Hata/Numano angiographic classification is widely used to categorize Takayasu arteritis into five types based on arterial involvement: Type I: Branches of the aortic arch Type IIa: Ascending aorta, arch, branches Type IIb: Type IIa + thoracic descending aorta Type III: Thoracic descending and abdominal aorta Type IV: Abdominal aorta and/or renal arteries Type V: Entire aorta and its branches. This classification correlates with clinical presentation and guides treatment strategies. Radiographic Features CT Angiography (CTA) CTA is the preferred modality for mapping vascular anatomy. It helps delineate the severity of stenosis, occlusions, aneurysms, and collateral pathways. In active disease, CTA reveals long-segment concentric mural thickening with homogeneous enhancement. In chronic stages, fixed luminal narrowing, post-inflammatory calcifications, and aneurysmal changes are evident. Characteristic CTA Signs: Double-ring sign: This sign features an inner low-attenuation ring within an enhancing vessel wall, correlating with mural edema and inflammation. Diffuse narrowing: This is observed in both the thoracic and abdominal aorta, particularly with ostial stenoses of branch vessels like the renal and mesenteric arteries. Collateral development: This occurs in chronic disease, providing indirect evidence of long-standing vascular compromise. MRI and PET/CT MRI is useful for detecting mural edema and enhancement on vessel wall imaging. FDG-PET/CT can demonstrate increased metabolic activity in inflamed vessels. These modalities are superior to CTA for monitoring disease activity and guiding immunosuppressive therapy, as emphasized in EULAR guidelines. Treatment and Prognosis High-dose corticosteroids remain the first-line therapy. They are often combined early with steroid-sparing immunosuppressants. Tocilizumab and other biologics have shown efficacy in refractory cases. Surgical or endovascular revascularization is reserved for severe, flow-limiting lesions and is ideally performed when inflammation is controlled. Relapses are common, making long-term follow-up with multimodality imaging essential to monitor disease progression and therapeutic response. Conclusion Understanding Takayasu arteritis is crucial for timely diagnosis and effective management. By recognizing the clinical features and utilizing appropriate imaging techniques, we can improve patient outcomes. References Rutter M, Bowley J, Lanyon PC, Grainge MJ, Pearce FA. A systematic review and meta-analysis of the incidence rate of Takayasu arteritis. Rheumatology (Oxford). 2021;60(11):4982-4990. doi: https://doi.org/10.1093/rheumatology/keab406 . Kerr GS, Hallahan CW, Giordano J, et al. Takayasu arteritis. Ann Intern Med. 1994;120(11):919-929. doi: https://doi.org/10.7326/0003-4819-120-11-199406010-00004 . Hata A, Noda M, Moriwaki R, Numano F. Angiographic findings of Takayasu arteritis: new classification. Int J Cardiol. 1996;54 Suppl:S155-S163. doi: https://doi.org/10.1016/S0167-5273(96)02813-6 02813-6). Dejaco C, Ramiro S, Duftner C, et al. EULAR recommendations for the use of imaging in large vessel vasculitis in clinical practice. Ann Rheum Dis. 2018;77(5):636-643. doi: https://doi.org/10.1136/annrheumdis-2017-212649 . Matsunaga N, Hayashi K, Sakamoto I, Ogawa Y, Matsumoto T. Takayasu arteritis: protean radiologic manifestations and diagnosis. Radiographics. 1997;17(3):579-594. doi: https://doi.org/10.1148/radiographics.17.3.9153698 . Yamada I, Nakagawa T, Himeno Y, Numano F, Shibuya H. Takayasu arteritis: evaluation of the thoracic aorta with CT angiography. Radiology. 1998;209(1):103-109. doi: https://doi.org/10.1148/radiology.209.1.9769819 . Park JH, Chung JW, Im JG, Kim SK, Park YB, Han MC. Takayasu arteritis: evaluation of mural changes in the aorta and pulmonary artery with CT angiography. Radiology. 1995;196(1):89-93. doi: https://doi.org/10.1148/radiology.196.1.7784596 . Kim SY, Park JH, Chung JW, et al. Follow-up CT evaluation of the mural changes in active Takayasu arteritis. Korean J Radiol. 2007;8(4):286-294. doi: https://doi.org/10.3348/kjr.2007.8.4.286 . Zhu FP, Luo S, Wang ZJ, Jin ZY, Zhang LJ, Lu GM. Takayasu arteritis: imaging spectrum at multidetector CT angiography. Br J Radiol. 2013;85(1020):e1282-e1292. doi: https://doi.org/10.1259/bjr/25536451 . 10. Bois JP, Anand V, Anavekar NS. Detection of inflammatory aortopathies using multimodality imaging. Circ Cardiovasc Imaging. 2019;12(7):e008471. doi: https://doi.org/10.1161/CIRCIMAGING.118.008471 . 11. Nakaoka Y, Isobe M, Takei S, et al. Efficacy and safety of tocilizumab in patients with refractory Takayasu arteritis: results from a randomized, double-blind, placebo-controlled, phase 3 trial in Japan (the TAKT study). Ann Rheum Dis. 2018;77(3):348-354. doi: https://doi.org/10.1136/annrheumdis-2017-211878 . 12. Hellmich B, Agueda A, Monti S, et al. 2018 update of the EULAR recommendations for the management of large-vessel vasculitis. Ann Rheum Dis. 2020;79(1):19-30. doi: https://doi.org/10.1136/annrheumdis-2019-215672 . Kevin M. Rice, MD is the president of Global Radiology CME and is a radiologist with Cape Radiology Group . He has held several leadership positions including Board Member and Chief of Staff at Valley Presbyterian Hospital in Los Angeles, California. Dr. Rice has made several media appearances as part of his ongoing commitment to public education. Dr. Rice's passion for state-of-the-art radiology and teaching includes acting as a guest lecturer at UCLA. In 2015, Dr. Rice and Natalie Rice founded Global Radiology CME to provide innovative radiology education at exciting international destinations, with the world's foremost authorities in their field. In 2016, Dr. Rice was nominated and became a semifinalist for a "Minnie" Award for the Most Effective Radiology Educator. He was once again a semifinalist for a "Minnie" for 2021's Most Effective Radiology Educator by AuntMinnie.com . He has continued to teach by mentoring medical students interested in radiology . Everyone he has mentored has been accepted into top programs across the country, including Harvard, UC San Diego, Northwestern, Vanderbilt, and Thomas Jefferson. Follow Dr. Rice on Twitter @KevinRiceMD All posts by Kevin M. Rice, MD